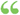 C2H2 1,493-4 The values first given above are based on the determination of calorific power made by Berthelot as given in Poole's "Calorific Power of Fuels," and the weight of gases calculated from the weight of air as determined by Regnault and afterward... C2H2 1,493-4 The values first given above are based on the determination of calorific power made by Berthelot as given in Poole's "Calorific Power of Fuels," and the weight of gases calculated from the weight of air as determined by Regnault and afterward...  Proceedings of the American Gas Light Association ... - Էջ 125American Gas Light Association - 1903Ամբողջությամբ դիտվող Proceedings of the American Gas Light Association ... - Էջ 125American Gas Light Association - 1903Ամբողջությամբ դիտվող - Այս գրքի մասին
 | American Gas Institute - 1907 - 1050 էջ
...Fuels," and the weight of gases calculated from the weight of air as determined by Regnault and afterward corrected for slight errors. By the calorific or heating...calorific value is ordinarily expressed in heat units per cub)c foot. In the case of liquids and solids, the heating value is expressed in heat units per pound.... | |
 | American Gas Institute - 1907 - 1050 էջ
...Fuels," and the weight of gases calculated from the weight of air as determined by Regnault and afterward corrected for slight errors. By the calorific or heating...the total number of heat units that are developed b\- the complete oxidation of all the combustible materials contained in unit weight or unit volume... | |
 | American Gas Institute - 1908 - 1166 էջ
...power, or calorific value, of a substance ? How is the heating power of a substance determined? Ans. By the calorific or heating value of a fuel is meant...ordinarily expressed in heat units per cubic foot. Thus hydrogen has a heating value of 326.2 Btu (British thermal units) per cubic foot. In other words,... | |
 | Wallace Ludwig Lind - 1920 - 240 էջ
...carbon and oxygen, so that fuels rich in hydrogen have high " calorific " values. By the calorific value of a fuel is meant the total number of heat units evolved during the complete combustion of unit weight (1 Ib.) of the fuel. In the following considerations... | |
 | Arthur William Judge - 1921 - 694 էջ
...in hydrogen should have high " Calorific " values. Here it should be explained that by the calorific value of a fuel is meant the total number of heat units evolved during the jombustion of unit weight of the fuel. In the following considerations the atomic... | |
| |